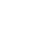H2(g)= H2(a)=2H(a)=2H(s) (17-1)
Here, g, a, and s denote gaseous hydrogen, adsorbed hydrogen, and dissolved hydrogen, respectively.
Pressure has a negligible effect on the equilibrium between solid and liquid phases, but is an important factor for reactions involving a gas phase. For instance, at a given temperature, the maximum solubility of a gas in a metal increases significantly with rising gas pressure. Thus, changes in pressure substantially alter the shape of gas–metal binary phase diagrams.
Under thermal equilibrium, the relationship between the hydrogen pressure P and the maximum solubility S of hydrogen in the metal ([H]/[M]) follows Sieverts' Law (for diatomic gases).